So32- lewis structure molecular geometry
The SO32- Lewis structure is an important concept in chemistry, as it helps us understand the molecular geometry of this particular ion. Molecular geometry refers to the arrangement of atoms in a molecule and the shape that the molecule takes as a result. Understanding the molecular geometry of a molecule or ion is crucial in predicting its physical and chemical properties. To understand the SO32- Lewis structure, lets first break down its components. The SO32- ion consists of one sulfur atom (S) and three oxygen atoms (O). The -2 charge on the ion indicates that it has gained two extra electrons, resulting in a total of 32 valence electrons. To determine the Lewis structure, we need to follow a few steps: Step 1: Calculate the total number of valence electrons. In this case, we have 32 valence electrons, which we can calculate by adding the valence electrons of each atom. Sulfur has six valence electrons (Group 16) and oxygen has six valence electrons (Group 16), so the total is 6 + 6 + 6 + 6 + 6 = 30. Step 2: Determine the central atom. The central atom is usually the least electronegative atom, and in this case, sulfur is the central atom. Sulfur is less electronegative than oxygen, making it the best choice for the central atom. Step 3: Connect the atoms. Next, connect the sulfur atom to each oxygen atom using a single bond (a line). Each bond consists of two electrons, which we can subtract from the total valence electrons. So, we have used four electrons from the total of 32, leaving us with 32 - 4 = 28 electrons. Step 4: Distribute the remaining electrons. The remaining 28 electrons need to be distributed around the atoms to satisfy the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons. In this case, sulfur and oxygen will be surrounded by eight electrons each. To distribute the electrons, we can place lone pairs around the atoms. Lone pairs are pairs of electrons that are not involved in bonding. Since oxygen is more electronegative than sulfur, it is more likely to have lone pairs. We can place two lone pairs on each oxygen atom, giving them eight electrons each. Step 5: Count the total number of electrons. After distributing the remaining electrons, count the total number of electrons to ensure that each atom has eight electrons (except for hydrogen, which only needs two). In this case, sulfur has eight electrons, and each oxygen atom has eight electrons. Step 6: Check the formal charges. Formal charge is a calculation used to determine the distribution of electrons in a molecule or ion. It is calculated by subtracting the number of lone pair electrons and half of the bonding electrons from the number of valence electrons. Each oxygen atom will have a -1 formal charge, and the sulfur atom will have a +2 formal charge. The final SO32- Lewis structure will have a central sulfur atom bonded to three oxygen atoms. Each oxygen atom will have two lone pairs, and the sulfur atom will have a +2 formal charge. The structure will have a bent or V-shaped molecular geometry, with the sulfur atom in the center and the oxygen atoms forming a triangle around it. In conclusion, the SO32- Lewis structure and its molecular geometry are crucial to understanding the properties and behavior of this ion. By following the steps outlined above, we can determine the correct Lewis structure, which helps us understand the arrangement of atoms and the shape of the molecule. The bent or V-shaped molecular geometry of SO32- is a result of the lone pairs on the oxygen atoms and the formal charges on the sulfur atom.
SO3 2- Molecular Geometry / Shape and Bond Angles - YouTube. A quick explanation of the molecular geometry of SO3 2- (Sulfite ion) including a description of the SO3 2- bond angles.. SO32- Molecular Geometry / Shape and Bond Angles (note so32- lewis structure molecular geometry. - YouTubegirl i want your physical chemical fuck then dont be
. A quick explanation of the molecular geometry of SO32- including a description of the SO32- bond angles so32- lewis structure molecular geometry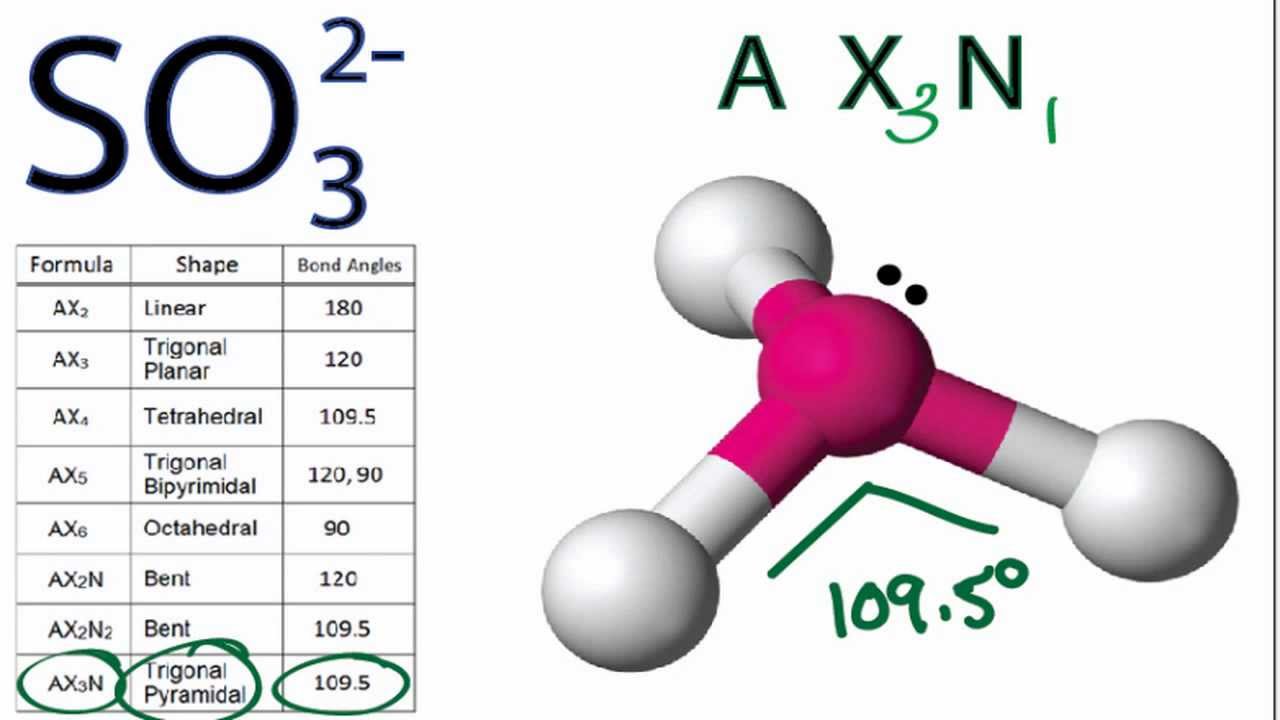
find local fuck friends without profile
. The central atom has 3 atoms and a lone pair and therefore, the electron geometry is tetrahedral while the molecular geometry is trigonal pyramidal. Steric number 4 corresponds to sp3 -hybridization where the idealized bond angles are 109.5 o.. What is the molecular geometry of SO3^2 - Toppr so32- lewis structure molecular geometry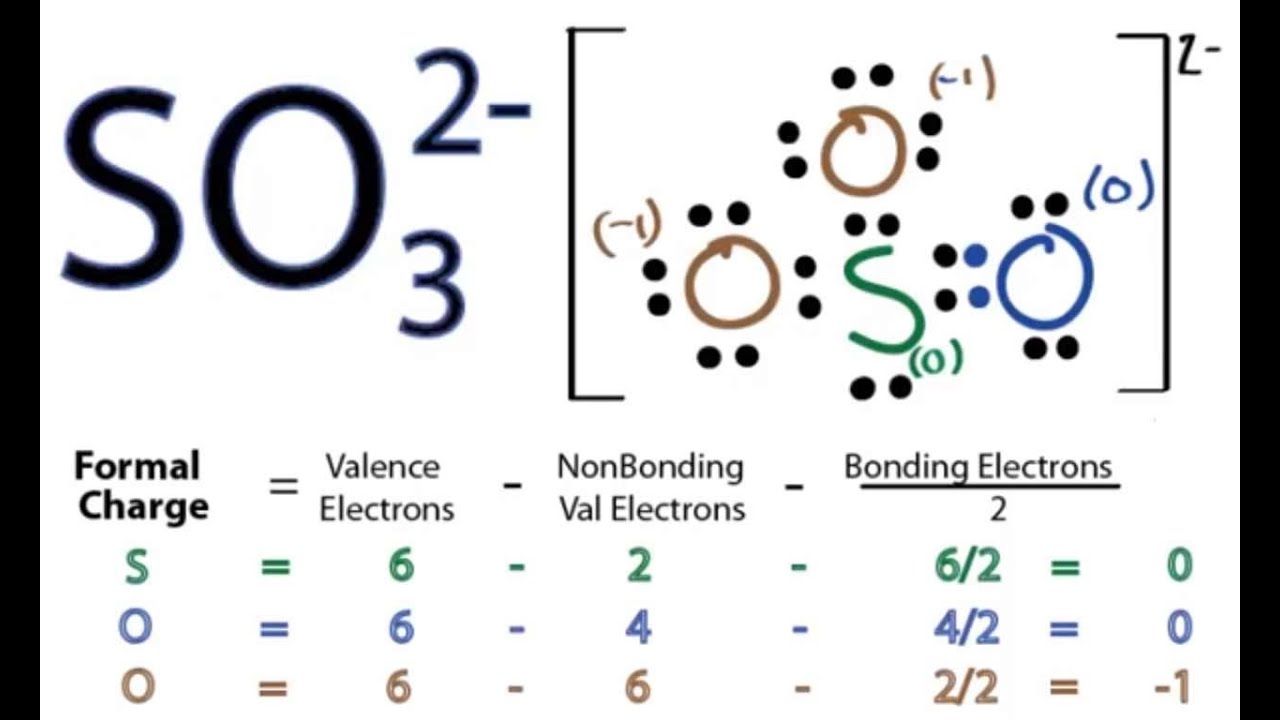

marketing giveaways
. draw - Chegg. Question: What is the molecular geometry of SO3 ^-2 a. draw its Lewis structure b. state its numeric code ci want to fuck a lesbian
. state its molecular geometry d so32- lewis structure molecular geometry. how many lone pairs of electrons are present on the central atom in the Lewis structure of sulfite ion? e. what is the formal charge on the central atom in sulfite ion? show calculations please f. how many polar covalent bonds. Draw the Lewis dot structure for SO32-. Determine the electron geometry .. 21K Learn what Lewis dot structures are, how to draw Lewis dot structures and see resonance in Lewis dot structures using the benzene Lewis dot structure example. Related to this.. SO3 Molecular Geometry, Lewis Structure, and Polarity Explained. Molecular geometry is the three-dimensional structure of the atoms which helps in the constitution of a molecule. It can determine reactivity, polarity, color, attraction, biological activity, etc so32- lewis structure molecular geometry. SO3 includes two components mainly - Sulfur and Oxygen. There are one sulfur atom and three oxygen atoms which are spread out as far away as they can! so32- lewis structure molecular geometry. SO42- lewis structure, molecular geometry, and hybridization - Topblogtenz. The Lewis structure of a sulfate [SO4]2- ion consists of 1 sulfur (S) atom and 4 atoms of oxygen (O). The sulfur atom is present at the center of the Lewis structure while the oxygen atoms occupy terminal positions so32- lewis structure molecular geometry. There are a total of 4 electron density regions around the central S atom in the Lewis structure of [SO4]2-.. SO3 2- Lewis Structure - How to Draw the Lewis Structure for SO3 2 .. Wayne Breslyn 631K subscribers Subscribe 1.7K 378K views 9 years ago A step-by-step explanation of how to draw the SO3 2- Lewis Structure (Sulfite Ion). For the SO3 2- Lewis structure.. Draw the Lewis structure for SiO32- with all resonance forms. State its .. Draw the Lewis structure for TeF4wwe.com universal sweepstakes
. For this molecule, determine the molecular geometry, electron domain geometry, bond angles, and hybridization about the central atomstop find sweepstakes
. Draw the Lewis structure and write the molecular geometry and hybridization on the central atom, and polar or nonpolar for SeCl_6.. Solved Question 1 From the Lewis Structure of SO32. sulfite - Chegg. How many resonance structures ? 6. What is the hybridization so32- lewis structure molecular geometry. Question: Question 1 From the Lewis Structure of SO32 so32- lewis structure molecular geometry. sulfite ion, answer the following questions 1. How many bonding pairs of electrons around the central atom? 2. How many nonbonding pairs of electrons around the central atom? 3.. Draw the Lewis structure for SO32- and provide the following .
mylabs ny wellnow

you sausage looking fuck
. Lewis structure is the distribution of the electrons around the atoms of a compound. This structure helps us to know about the kind of bonds and the number of bonds that form the compound so32- lewis structure molecular geometry. Now lets walk through the method of drawing lewis structure:. Solved . Draw the Lewis dot structure for the SO32 - Chegg
lesbian dating sims
. SO3 2- : Lewis Structure and Molecular Geometry - YouTube. Chemistry learning made easy. This tutorial will help you deal with the lewis structure and molecular geometry of sulfite ion (SO3 2-)i want to date my fuck buddy
. 3.8M views 254K views Almost yours: 2 weeks, on us.. Answered: For SO3^-2 provide a Lewis structure,… | bartleby. For SO3^-2. provide a Lewis structure, predicted VSEPR molecular geometry, bond angle and indicate whether the compound is polar, nonpolar, or a polyatomic ion..